 3.15.24 | Endometriosis
3.15.24 | Endometriosis Closing Gaps in Physician Endometriosis Education
While endometriosis impacts such a large group of individuals across the country, there are still significant gaps in provider education surrounding endometriosis diagnosis and treatment.
 3.8.24 | Policy
3.8.24 | Policy Statement from the Society for Women’s Health Research on President Biden’s 2024 State of the Union Address
March 7, 2024—The Society for Women’s Health Research released the following statement in response to the 2024 State of the […]
 3.6.24 | Menopause
3.6.24 | Menopause Menopause Resource Guides Aim to Create Menopause-Friendly Workplaces for All
Inspired by the EMPACT Menopause Study findings, SWHR developed a set of resource guides to help address these significant gaps for midlife and older women and ultimately help improve wellness for all individuals at work.
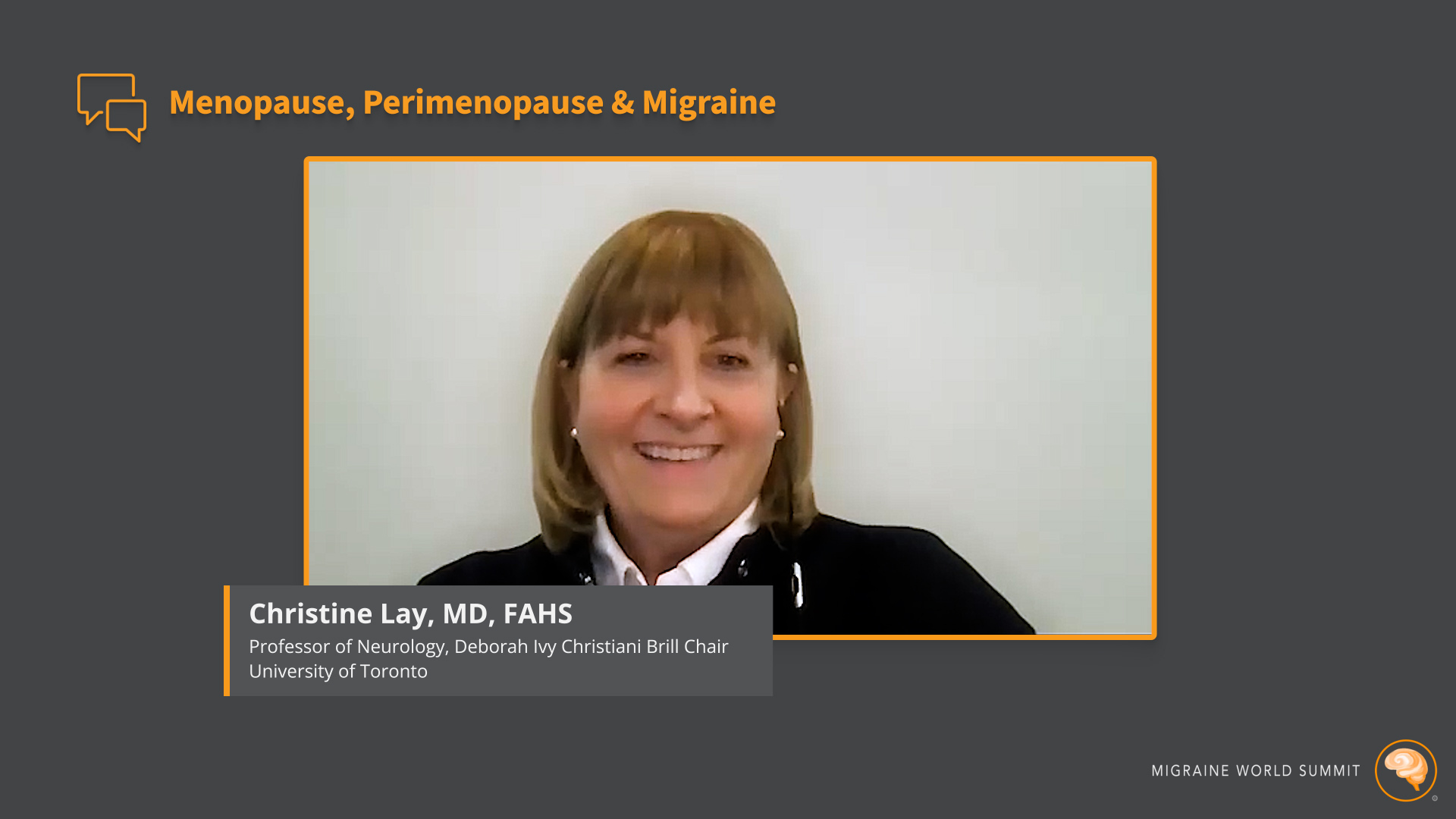 2.28.24 | Migraine
2.28.24 | Migraine Menopause, Perimenopause, and Migraine
During an interview for the 2024 Migraine World Summit, Dr. Christine Lay shared what she has discovered about menopause and migraine based on her medical practice.
 2.26.24 | Autoimmune
2.26.24 | Autoimmune Elevating the Impacts of Autoimmune Disease and Black Women’s Health
While it is unclear what makes someone more at risk for an autoimmune disease, constructs, such as race and ethnicity, and social determinants of health, including education level, income, and other socioeconomic measures, are known to have an impact on health outcomes.
 2.26.24
2.26.24 SWHR Welcomes Three New Members to Board in 2024
SWHR is pleased to welcome three new leaders Shyamasundaran Kottilil, MD, PhD, Christine Harhaj, and Catherine Spong, MD to its Board of Directors.
 2.23.24 | Heart Health
2.23.24 | Heart Health SWHR Launches Heart Health Policy Agenda
SWHR launched its latest policy agenda, "Improving Women's Heart Health Outcomes Across the Lifespan," informed by an interdisciplinary Heart Health Policy Working Group of policy professionals, researchers, clinicians, and patient advocates.
 2.21.24 | Policy
2.21.24 | Policy How SWHR is Driving its 2023-2024 Policy Agenda Priorities
SWHR launched its 2023-2024 Federal Legislative Agenda, outlining key areas for SWHR engagement in the legislative and regulatory arenas. The activities are categorized into Core Leadership Activities, Key Policy Priorities and Areas of Active Engagement, and Other Areas for Potential Engagement.
 2.13.24 | Cancer
2.13.24 | Cancer Cervical Cancer Awareness Month Event Spotlights How to Prevent HPV-Related Cancers
Read takeaways from the HealthyWomen’s Congressional Briefing on Preventing HPV-Related Cancers through Vaccination and Screening, in recognition of HPV Prevention Week and Cervical Cancer Awareness Month.
 2.7.24 | Health Equity
2.7.24 | Health Equity A Call to Action to Addressing Heart Health Across a Woman’s Lifespan, Informed by Data Dashboard
To offer solutions and bring attention to IHD and American Heart Month this February, SWHR has released a resource spotlighting actions to address gaps in heart health education for women across the lifespan.









